 
Where c = 2.998 x 10 8 m.s -1 (the velocity of light) and l is in meters.Į = h x 2.998 x 10 8 = 1.The base unit for energy is joules (Non-SI/Derived Unit) Most usually in photosynthetic work, l (wavelength) is used in place of n. Where h = 6.626 x 10 -34 J.s n is the frequency (s -1). Taking a=3 H +/ATP and Dp=0.24 volt, a typical value, then:Įnergy of a photon is given by Plank's equation: Where a is the overall stoichiometry of the proton pump. If a proton motive force ( Dp) is in equilibrium with a phosphorylation system in which the free energy for ATP synthesis is DG', then: Under conditions where DG' for ATP hydrolysis is say -60 kJ.mol -1, then If this energy was used to drive a process involving 2 charges (e.g., 2 e - or 2 H +):ĭG o' elec = - DG o' = 30 x 10 3 = 0.156 V When considering the proton gradient that can be driven by this work term, where Dp is expressed in V, it is useful to express this in electrical units. Thus the standard free energy of hydrolysis ( DG o') of ATP is approximately -30 kJ.mol -1. It is permissible and sometimes useful to express the energy of change of other processes in electrical units. Thus, if DE' = 0.1 volt, then for a reaction involving 2 equivalents: Here the convention determines that the redox change is spontaneous ( DG' is negative) for a reaction in which the oxidizing couple has a higher E' than the reducing couple. Where z is the number of redox equivalents involved in the reaction (usually z = 2 (for quinones, NAD+/NADH, etc.), or z = 1 (for most cytochromes)), and DE' means (E' (oxidizing couple) - E' (reducing couple)). For two redox couples differing in potential by an amount DE', the free energy per mol which is available from electron transfer between them is given by: The Farady constant provides a factor relating electrical potential difference to free energy. The units for RT/F are obtained as follows: One singly charged species (an electron or H +, for example) has a charge of 1.602 x 10 -19 C, so this multiplied by Avagadro's constant is the faraday (F). So C has base units of amp.s and F has base units of amp.s. The number of entities in a mole is 6.022 x 10 23, which is Avagadro's constant, N.Įlectrical charge: A mole of singly-charged species has a charge of 9.64853415 x 10 4 C (the faraday, F, rounded to 9.649 x 10 4 C in this course) a Coulomb (C) is the charge carried by a current of one of the ampere in one second. Entities may be atoms, molecules, ions, electrons, or other particles or groups of particles. Substance: the amount of a substance is measured by the mole (mol) the mole is defined as the amount of substance of a system which contains as many entities as there are atoms in 0.012 kg of the carbon isotope 12 6C. Since the derived unit of a volt is defined with units W.A -1, and the derived unit of a watt (W) is defined in J.s -1, we get overall A.J.s -1.A -1.s, giving J = A.V.s. Work is given by (w = force x distance) since force = mass x acceleration, with base units kg.m.s -2 (defining the derived unit of a newton (N)), 1 joule = N.m with base units of kg.m 2.s -2.Įlectrically, a joule is equivalent to the energy dissipated by a current of one ampere (A) passing though a potential of one volt (V) for one second. Mechanically, a joule is equivalent to the energy dissipated by a force of one newton acting through 1 meter. 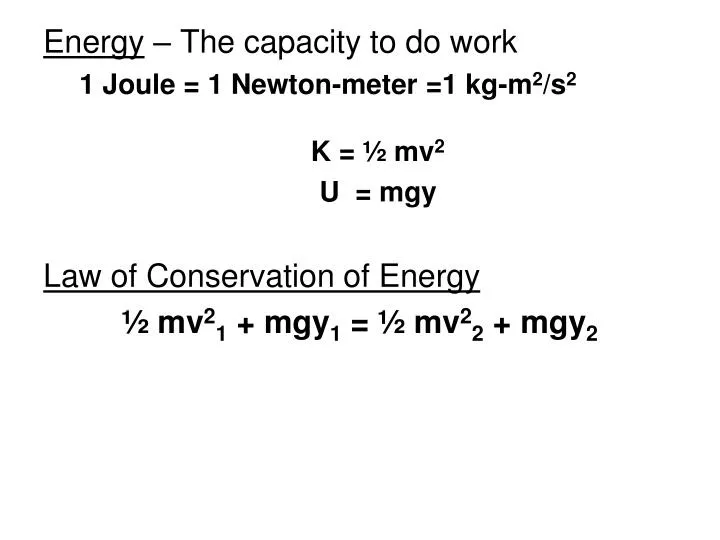
Untitled UNITS Energy (or work ): E (or w) is expressed in joules (J) or calories (cal). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
